When you pick up a prescription, you might not think twice about the pill’s color, the bottle label, or the company name printed on it. But if you’re trying to save money or understand what you’re really taking, those details matter - especially when it comes to authorized generics.
Authorized generics aren’t just another type of generic drug. They’re the exact same medicine as the brand-name version, made in the same factory, with the same ingredients, and the same effectiveness. The only differences? The packaging and the label. And that’s where things get confusing.
What Makes an Authorized Generic Different?
Most generic drugs are approved through the FDA’s Abbreviated New Drug Application (ANDA) process. That means they must prove they’re bioequivalent to the brand drug - meaning they work the same way in your body. But authorized generics skip that step entirely. They’re made by the original brand manufacturer or under its direct license, using the same formula, same equipment, and same quality controls. The FDA defines them as a listed drug marketed under the original New Drug Application (NDA), just with different packaging.
Think of it like this: The brand-name drug is sold in a blue bottle with the company logo. The authorized generic? Same pill, same strength, same everything - but it’s in a plain white bottle with a different company name on it. No marketing. No fancy branding. Just the medicine.
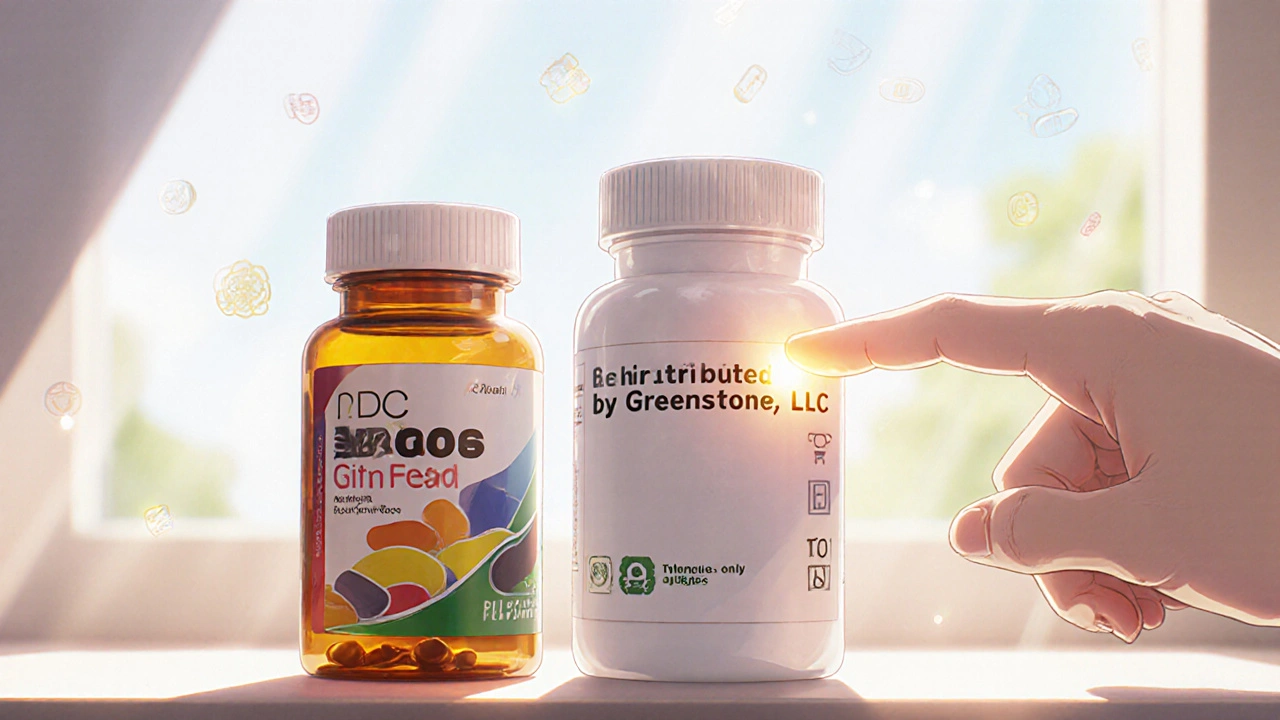
How to Spot an Authorized Generic by the Label
Here’s how to tell an authorized generic from the brand version or a regular generic:
- No brand name on the label - If you see "Pfizer" or "AstraZeneca" on the bottle, it’s the brand. If you see "Greenstone LLC" or "Dr. Reddy’s Laboratories," it’s likely an authorized generic.
- Different labeler code in the NDC number - Every drug has a National Drug Code (NDC), a 10-digit number broken into three parts. The first part is the labeler code - who’s responsible for the product. The brand and its authorized generic will have the same product code and package code, but different labeler codes. For example, Protonix (brand) might have NDC 00071-0001-01, while its authorized generic is 55111-0001-01. Same drug, different distributor.
- "Distributed by" instead of "Manufactured by" - Authorized generics often say "Distributed by [Company Name]" on the label. The actual manufacturer is usually the same as the brand, but that’s not always printed.
- No trademark symbols (® or ™) - You won’t see the brand’s trademark on authorized generics. The labeling is stripped down to just the facts: drug name, strength, dosage, and manufacturer info.
Some authorized generics even have a small "AG" or "Authorized Generic" printed in tiny text on the bottom of the label - but this isn’t required by the FDA. Don’t rely on it.
Why Authorized Generics Look So Much Like the Brand
It’s not a mistake. It’s by design.
When a brand-name drug’s patent expires, the first company to file a generic application gets 180 days of market exclusivity. During that time, no other generics can enter. But the brand company doesn’t want to lose its customers. So they launch their own authorized generic - same pill, different label - to compete with the first generic. That keeps their market share and gives patients a lower-cost option.
Because they’re made in the same facility, the pills often look identical. Same color, same shape, same imprint code. That’s why so many people think they’re the same product. They are - just sold under a different name.
How Authorized Generics Differ From Regular Generics
Not all generics are created equal. Here’s how authorized generics stack up against traditional ones:
| Feature | Authorized Generic | Traditional Generic |
|---|---|---|
| Manufacturer | Same as brand-name drug | Separate company |
| Active Ingredients | Identical | Identical |
| Inactive Ingredients | Identical | May differ |
| Approval Pathway | Under original NDA | Through ANDA |
| NDC Labeler Code | Different from brand | Completely different NDC |
| Labeling Updates | Auto-updated with brand | Must be manually updated |
Because authorized generics share the same NDA as the brand, they automatically get any safety updates or label changes the brand receives. Traditional generics have to submit their own updates - and sometimes those updates get delayed.
How to Verify an Authorized Generic
Don’t guess. Check.
The FDA publishes a Quarterly Authorized Generic List - updated every January, April, July, and October. As of October 2023, it includes 147 authorized generic products across 68 drug families. This is the only official source.
Here’s how to use it:
- Find the NDC number on your prescription bottle.
- Look up the first segment (labeler code) on the FDA’s list.
- If it matches a listed authorized generic, you’ve got one.
Most pharmacy systems - like First Databank and Medi-Span - now flag authorized generics automatically. But if you’re unsure, ask your pharmacist. They can cross-check the NDC against the FDA list in seconds.
Why This Matters to You
Authorized generics cost less than the brand - often 15% to 25% lower - but they’re usually a bit more expensive than traditional generics. Still, they offer a middle ground: the reliability of the original manufacturer without the brand-name price tag.
Patients who switch from brand to authorized generic rarely report any change in how the drug works. In a 2022 Medscape survey, 92.6% of patients said they noticed no difference in effectiveness or side effects.
But confusion is common. A 2022 NCPA survey found that 63% of pharmacists spent over two minutes per prescription verifying whether a drug was an authorized generic. That’s because the packaging is so close to the brand. Some patients even think the different label means it’s fake or inferior.
It’s not.

Common Mistakes and How to Avoid Them
Here are the top three errors people make:
- Thinking different packaging = different drug - Same pill, different bottle. That’s all.
- Confusing authorized generics with "authorized brand" products - Some companies repackage traditional generics in brand-like packaging to trick people. These aren’t authorized generics. They’re just clever marketing.
- Assuming all generics are the same - Authorized generics are closer to the brand than traditional generics in every way - including inactive ingredients and labeling updates.
If you’re unsure, always ask your pharmacist to confirm the NDC against the FDA list. You have the right to know exactly what you’re taking.
What’s Changing in 2024
The FDA plans to integrate authorized generic identifiers directly into the National Drug Code Directory by Q2 2024. That means pharmacy systems will flag them automatically - no more manual checks. It’s a step toward reducing dispensing errors, which currently account for 8.3% of generic drug mistakes, according to the ISMP National Medication Errors Reporting Program.
Meanwhile, authorized generics are growing. They made up 12.7% of the generic drug market by revenue in 2022 - up from 9.5% in 2020. That growth is especially strong in specialty drugs, where cost savings matter most.
As more brand companies use authorized generics to stay competitive, knowing how to spot them will become even more valuable.
Are authorized generics as safe as brand-name drugs?
Yes. Authorized generics are made in the same facility, with the same ingredients, and under the same quality controls as the brand-name drug. The FDA inspects these facilities just like it does for brand-name manufacturers. There’s no difference in safety or effectiveness.
Why do authorized generics have different NDC numbers?
The NDC has three parts: labeler code, product code, and package code. The labeler code identifies who’s responsible for distributing the drug. Authorized generics are distributed by a different company (often a subsidiary), so they get a new labeler code. But the product and package codes stay the same - meaning it’s the exact same drug.
Can I trust an authorized generic if the label looks different?
Absolutely. The FDA requires authorized generics to have the same active ingredients, strength, dosage form, and route of administration as the brand. The only allowed differences are packaging, labeling, and distributor name. If the pill looks the same and the NDC matches the FDA list, it’s trustworthy.
Do authorized generics have the same side effects as the brand?
Yes. Because they contain identical active and inactive ingredients, side effects are the same. In fact, patients report fewer issues switching to authorized generics than to traditional generics, because there’s no change in formulation.
How do I know if my pharmacy is giving me an authorized generic?
Ask for the NDC number on the bottle and check it against the FDA’s Quarterly Authorized Generic List. You can also ask your pharmacist directly: "Is this an authorized generic?" Most pharmacy systems now flag them, but it never hurts to confirm.
What to Do Next
If you’re paying full price for a brand-name drug, ask your doctor or pharmacist: "Is there an authorized generic available?" It could save you money without changing how the medicine works.
Keep the FDA’s Authorized Generic List handy - print it or bookmark it. And next time you get a prescription, take a second to look at the label. That small detail might be the difference between paying too much - and getting the same medicine at a better price.
